
Medical Innovations Incubator
HOME OF 4C - Enabling Medical Innovations
Unser Fokus: Medizininnovationen
Wir fördern Medizininnovationen. Seit 2014 verfolgen wir gemeinsam mit unserer gemeinnützigen Muttergesellschaft, der Stiftung für Medizininnovationen, dieses große Ziel. Dabei sind uns die Herausforderungen der Gesundheitsbranche stets bewusst. Sie bilden das Fundament unseres täglichen Handelns in der Zusammenarbeit mit Startups und bereits etablierten Unternehmen mit innovativen Medizinprodukten und In-vitro-Diagnostika.
Unsere Erfahrung zeigt: Gerade für Gründer:innen sind Kenntnisse und Fähigkeiten der Entrepreneurship Essentials und gängige Methoden, wie Business Model Canvas, Design Thinking oder Lean Launchpad, zwar unerlässlich, um erfolgreich zu werden, benötigen Medtech-Startups jedoch zusätzliches Knowhow. Vor ähnlichen Herausforderungen stehen auch bereits etablierte Unternehmen, die eine Innovation außerhalb des aktuellen strategischen Fokus auf den Markt bringen möchten, sowie Unternehmen, die in dieser Branche ein neues Geschäftsfeld entwickeln möchten.
In den letzten Jahren haben wir daher unseren einzigartigen Ansatz entwickelt: Durch das 4C Konzept und der 4C Business Modelling-Methode enablen wir Medizininnovationen! Dieser Ansatz dient als Ausgangspunkt unserer vielfältigen Angebote für Startups und Corporates, aber auch für die Zusammenarbeit mit Investor:innen und der Wissenschaft.
Unsere 4Cs – Die Basis für unser Business Model
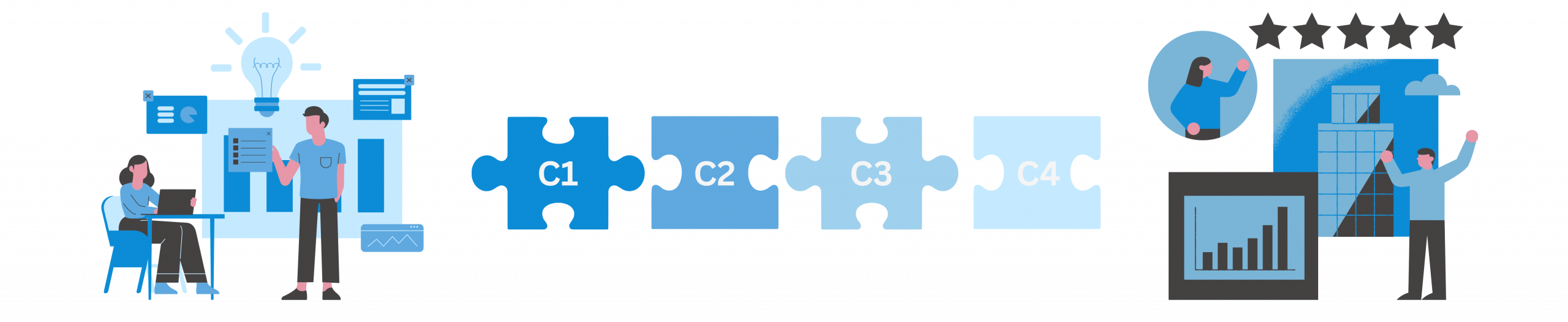
Wir vom Medical Innovations Incubator (MII) haben uns auf die gezielte Unterstützung von MedTech-Innovationen in den Bereichen Medizinprodukte, In-Vitro-Diagnostika und Digital Health spezialisiert. Hierfür haben wir einen ganzheitlichen Beratungsansatz entwickelt und betrachten die Herausforderungen und Chancen des Gesundheitsmarktes vernetzt. Unser Ansatz basiert auf den 4Cs: C1 | Commercialization, C2 | Certification, C3 | Clinical Studies und C4 | Copyright. Diese 4Cs bilden die Basis des Geschäftsmodells eines jeden Medtech-Unternehmens.
Hinter jedem unserer 4Cs stehen Anforderungen unterschiedlichster Interessengruppen. Besonders zentral sind dabei die Anforderungen der regulierten Gesundheitsbranche, zu der Gesetze, Normen und die zuständigen Behörden gehören. Gleichzeitig beeinflussen sich die einzelnen Cs gegenseitig, was ihre Komplexität weiter erhöht.
Was wir unter Enabling verstehen
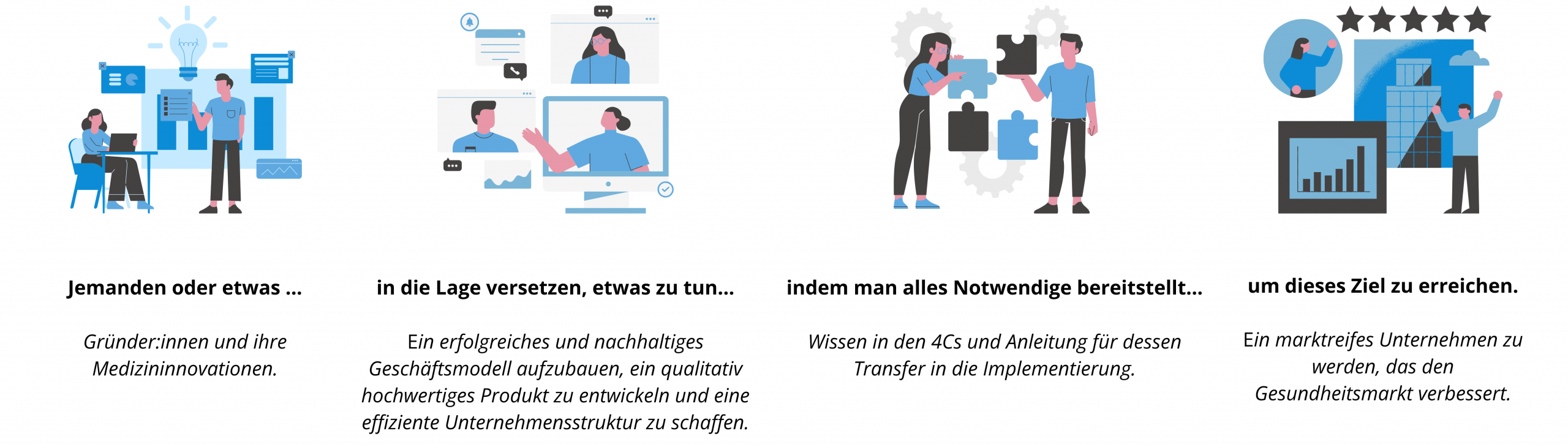
Unser Team aus Enabler:innen unterstützt euch dabei, die Dinge selbst in die Hand zu nehmen. Ganz nach dem Motto: Hilfe zur Selbsthilfe. All unsere Services – in Form von Beratung, Workshops, Reviews und Template-Packages – wurden deshalb didaktisch wertvoll und an die individuellen Bedürfnisse, Erfahrungen und Ressourcen jedes Teams anpassbar entwickelt.
